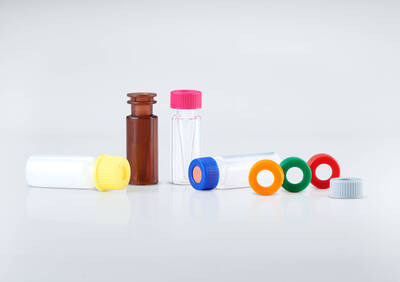
High-precision injection molding complete solutions for diagnostics and laboratory analytics.
Know-how
The correct diagnosis saves lives. Our drive for an even more precise measurement result.
Ready for new applications and opportunities.
The diagnostics industry is always moving. New trends and developments such as point-of-care immediate diagnosis systems (quick tests), micro-structured surfaces, glass replacement or increasingly smaller disposables with high throughputs require clear quality orientation along the entire added value chain. This means for us as a system supplier to exceed the normative requirements of our customers with flexible manufacturing processes, high-cavity tools and innovative product concepts. The reliability and stability of the process are always the priority for all economic considerations.
Production conditions
- Clean room ISO 7, DIN EN ISO 14644-1
- Hygiene monitoring
- Production with reduced ATP / RNA / DNA
- Certified quality management according to DIN EN ISO 13485
- GMP standard
Highest standardized quality and hygiene standards based on validated processes and a certified QM system.
Solutions and technology
Millions of units at constant high quality – not the only requirement for your disposable products and measuring systems.

Diagnostics complete solutions from the clean room
Based on consistent project and quality management, we process high quality disposable materials using the latest injection molding and assembly technologies. Multi-cavity tools with up to 96 cavities, 100% camera inspections, monitoring tests and highly automated assembly processes enable the production of high volumes with stable quality in a controlled clean room environment.
- Development and design
- Prototyping of demonstration and function samples
- Toolmaking
- Precision injection molding and extrusion blow molding
- Semi-automatic and completely automated clean room assembly, 100 % camera monitoring
- Sterilization and finishing services
- Automated packaging and logistics
The complete range in the diagnostics applications field.
We have already brought numerous diagnostics applications to series production in more than 30 years of industry expertise. Disposable system and functional components that prove themselves millions of times daily in the patient environment or laboratory use.